The U.S. Department of Health and Human Services (HHS) has tasked AktiVax, Inc. with developing an auto-injector that allows drugs designed to counter chemical nerve agents to be administered without specialized training.
The Biomedical Advanced Research and Development Authority (BARDA) awarded AktiVax of Boulder, Colo., an initial 18-month, $15 million contract to develop an auto-injector for drugs that counter the effects of organophosphates, a chemical class that includes agents like Sarin and VX. The contract could be extended to up to four years and $55 million.
“Chemical agents can kill within hours,” said Rick Bright, director of BARDA, which is administered through the HHS Office of the Assistant Secretary for Preparedness and Response. “To save lives, we need products at the ready that allow local first responders to act immediately.”
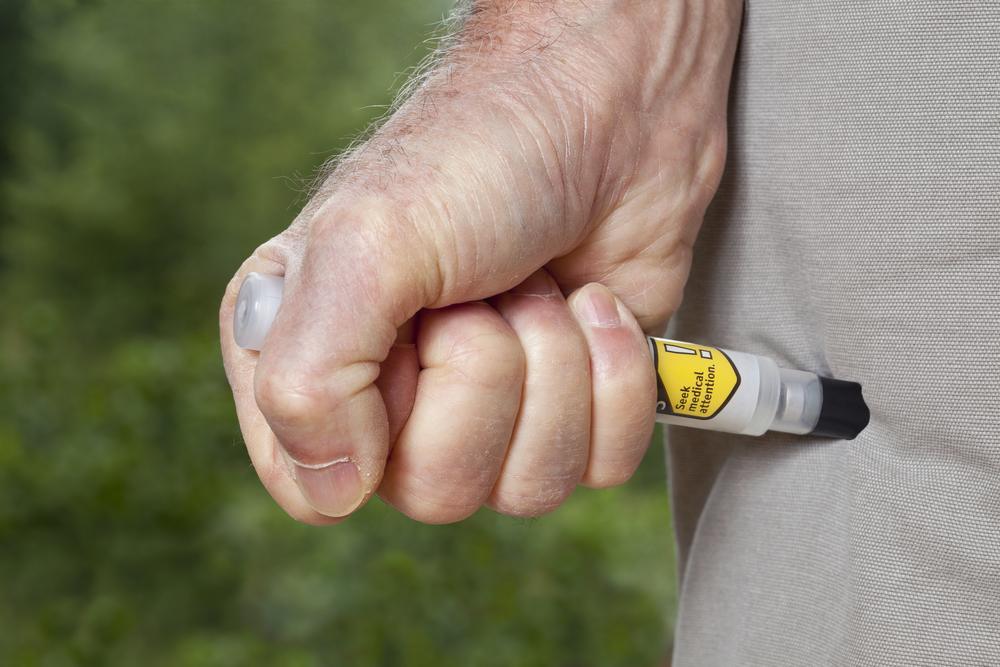
The federal government has stockpiled antidotes to nerve agents like 2-PAM and pralidoxime chloride. The drugs are administered with pre-filled auto-injector pens that are no longer in production.
Under the contract, AktiVax will work to modify its ARAI auto-injector platform so that it can be used to administer 2-PAM. The modified auto-injector could be used to administer additional drugs, or additional combinations, in the future.

